Boasting the fourth largest population in the world along with rapid growth of the middle class that has contributed to the consumption surge over the years, Indonesia makes an enticing destination for foreign investors engaging in the food and beverage (F&B) industry.
Furthermore, as a developing country, Indonesia requires more advanced technology in medical devices, thus demanding foreign supply. The healthcare industry, particularly the medical device sector, then is attractive to dive into.
IMPORTING FOOD AND BEVERAGES AND MEDICAL DEVICES INTO INDONESIA: THE ESSENTIALS
Basic Requirements
The basic requirements for registering F&B and medical devices in Indonesia are 90 percent the same. When submitting an online application, you are required to submit the following documents altogether:
- Company Documents: Single Business Identity Number (NIB), Company’s Tax ID (NPWP), Trade Business Licence (SIUP), Importer Identification Number (API), plus Medical Device Distribution Licence (IPAK) for medical devices.
- Administrative Documents: Letter of Authorisation (LoA), Free Sales Certificate, GMP Certificate/ISO.
- Technical Documents: Dossier from Manufacturer.
How to Secure an Import Licence
To streamline the process of securing an import licence that previously could take up to five months, the government has implemented the Online Single Submission (OSS) system since July 2018.
All business entities are required to register through the OSS. Once completed, an NIB is automatically issued. The NIB serves as the basic Import Licence and replaces Company Registration Certificate (TDP) and Importer Identification Number (API).
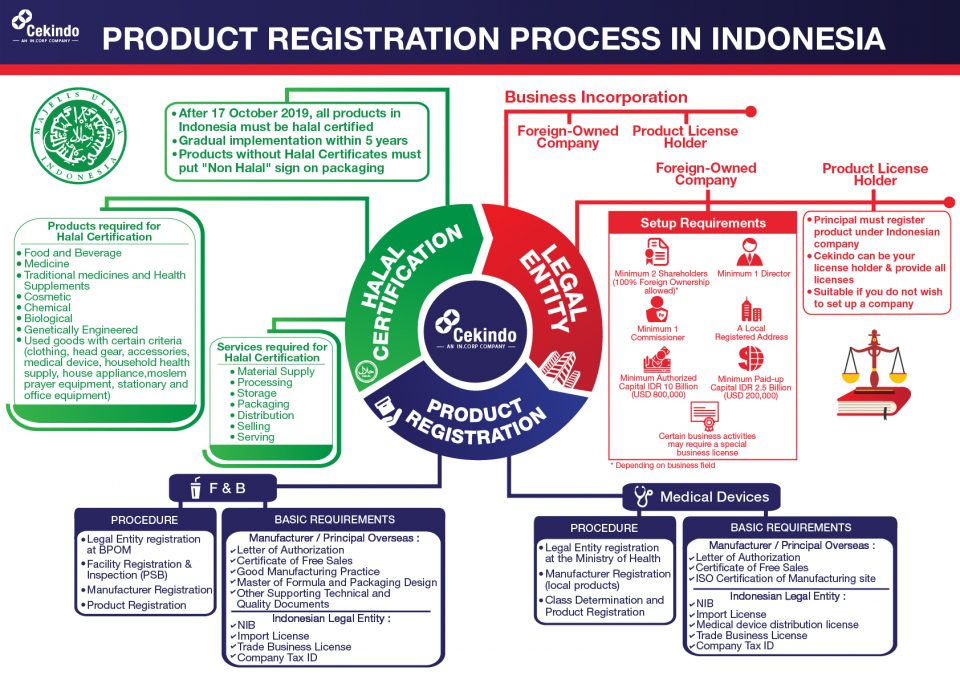
Procedures for Food and Beverage Registration
The procedures between registering F&B products and medical devices are slightly different.
In short, business owners are to go through the following procedures for successful F&B registration in Indonesia:
- Registering a legal entity (company) at BPOM system: carried out online.
- Registering the facility (warehouse) followed up by facility inspection.
- Registering manufacturer: carried out online.
- Registering products: products can finally be distributed and sold after getting product licence number (valid for five years).
In general, each F&B product must be registered. However, the government has made some exceptions. If a product is categorised as one of the following, it can be exempted from product registration:
- Used for personal consumption (with amount limit)
- Sold as ingredients to the manufacturer and not sold directly to end customer
- Short-lasting (its life cycle does not last more than seven days)
- Fast food
Different packaging material and design would have a separate registration. However, if they come in various sizes and weight, one registration is sufficient. For example, chocolate cookies that are to be sold in small and big packaging.
Procedures for Medical Device Registration
As for medical device registration, the following procedures are to be undertaken:
- Registering a legal entity (company) at the Ministry of Health: carried out online.
- Registering manufacturer (local products): MoH inspection to the local manufacturer.
- Registering products after device class determination: products can finally be distributed and sold after getting a medical device licence (valid for five years or according to the validity of LoA).
Indonesia implements a four-grade risk evaluation system, when medical devices are mishandled or not used properly:
- Class A (low risk): not harmful to humans.
- Class B (low to moderate risk): can have some serious effects but will not be considered as a serious accident to humans.
- Class C (moderate to high risk): can have very serious effects but will not be considered as a serious accident to humans.
- Class D (high risk): can have some serious effects and will be considered as a serious accident to humans.
COVID-19: Medical Device Licensing Requirements are being Relaxed
In an effort to battle the coronavirus and prevent its quick spread in Indonesia, the medical device licensing requirements for importation and distribution have been relaxed by the government, including a one-day service to get Distribution Permit (Izin Edar) for local products.
Eligible medical devices are those used for taking care of the COVID-19 pandemic, such as surgical gloves and apparel, ventilators, hand sanitizers, and surface or room disinfectants.
PT PMA ESTABLISHMENT BEFORE PRODUCT REGISTRATION IN INDONESIA
Since only companies that are legally registered in Indonesia with valid licences are allowed to register either food, beverages or medical devices, company establishment is required to proceed to product registration.
By law, foreign investors can establish a foreign-owned company (PT PMA). Depending on the business classification, a PT PMA allows up to 100% foreign ownership. An import-export company can be fully owned by foreigners, whereas a foreign distributor company can own 67 percent shares and a retailer of food and beverage products cannot be owned by foreigners.
Taking between one and 1.5 months to set up, a PT PMA requires an investment plan of Rp10 billion, with the paid-up capital of Rp2.5 billion.
ALTERNATIVES TO PT PMA SETUP IN INDONESIA
Foreign investors who are excited about starting business operations right away in Indonesia can start importing without having to establish a PT PMA and secure an import licence. These alternatives also serve as the ideal solution for foreign ownership restrictions when establishing a PT PMA.
Product Licence Holder
A Product Licence Holder, such as Cekindo, will provide importers with all necessary licences. All products that are to be imported will be registered based on a licence holder agreement without the right of exclusivity, meaning flexibility to engage several distributors is granted.
Importer of Record
Importer of Record (IOR), also known as Undername Importer, is the ideal solution to complete the product importation. An IOR is a registered legal entity with an import licence is responsible for taking care of all documents and entry requirements for importing goods into Indonesia.
SHOULD YOU CARE ABOUT HALAL CERTIFICATION?
Among the 270 million people in Indonesia, approximately 88 percent are Muslims, making the country hold the status of having the world’s largest Muslim population. Referring to the staggering percentage, investors should certainly care about halal certificates.
Furthermore, the government aims at transforming Indonesia into a producer of halal products and services, instead of simply becoming a market through the implementation of the sharia economy master plan.
Starting from October 2019, these product categories are required to have halal certificates from the BPJPH: food and beverages, drugs, traditional medicines and health supplements, cosmetics, chemical, biological, genetically engineered products, and used goods with certain criteria.
To secure a halal certificate, here are the steps:
- Implementing Halal Assurance System (HAS) of SNI 99001:2016.
- Preparing all required documents.
- Filling out documents according to certification status and submit to a representative.
- Guidance is provided for all audits and lab analysis by the representative.
- Halal Certificate is issued when the product meets the HAS requirements.
CONSULT A PROFESSIONAL
Complying with the local regulations of product registration, import-export and company establishment may be challenging to investors. For updates on regulation changes pertaining to COVID-19 situation and a step-by-step guide to ensure seamless importation and product registration in Indonesia, consult a reliable business consultant.
Visit www.cekindo.com/indonesia-expat-import for a free consultation. In addition to food and beverages and medical devices, we can assist in registering food supplements and cosmetics.
As part of InCorp Group in Singapore, Cekindo is a leading consulting company in Indonesia and Vietnam providing integrated market-entry and corporate services. Become one of our satisfied clients, jumpstart your business now.